Recently, a research team led by Professor Minghua Huang from the College of Materials Science and Engineering at Ocean University of China (OUC) has made significant advances in the field of non-precious metal-based oxygen electrocatalysts. The related results have been published in leading international journals including Advanced Energy Materials, Advanced Functional Materials, and ACS Catalysis. The research was supported by the General Program of the National Natural Science Foundation of China, the BRICS Science, Technology and Innovation Framework Program, and funding from central universities.
The development of efficient and durable non-precious metal-based oxygen electrocatalysts is essential for advancing next-generation energy devices, such as anion exchange membrane alkaline electrolyzers and metal–air batteries. However, the sluggish kinetics of oxygen reduction and evolution reactions (ORR/OER) significantly limit overall energy conversion efficiency. Therefore, designing high-performance catalysts that can replace commercial noble-metal catalysts (e.g., Pt/C and Ir/C) is crucial for the large-scale commercialization of these technologies.
Focusing on the unclear reaction mechanisms of dual-atom catalysts in ORR, the research team successfully designed and constructed alkaline earth metal calcium-based dual-atom catalysts (Ca₂–N–HCS) and transition metal FeNi dual-site catalysts (Fe–Ni–N–C). In the alkaline earth system, the study revealed that p–p orbital hybridization between Ca 3p orbitals forms a unique dual-channel electronic regulation mechanism featuring both σ-donation and π-backdonation (Fig. 1). This mechanism enhances Ca 3p–O 2p orbital hybridization, stabilizes Ca–O bonds, raises the p-band center, and precisely tunes the adsorption energies of oxygen intermediates, thereby accelerating ORR kinetics. Benefiting from this strategy, the catalyst demonstrates excellent durability and operational stability in zinc–air batteries over a wide temperature range from −40 °C to 60 °C.
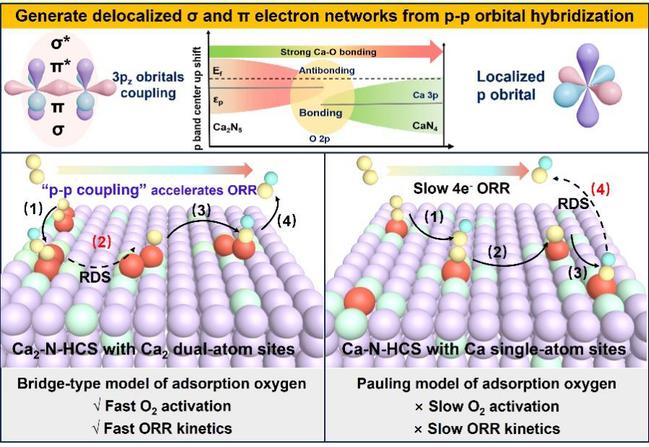
Figure 1 Schematic diagram of the mechanism of p-p orbital coupling to enhance ORR activity in alkaline earth metal calcium-based diatomic catalyst (Ca2–N–HCS).
Furthermore, compared with p-orbital-dominated alkaline earth systems, transition metal-based dual-atom catalysts (Fe–M–N–C, M = Mn, Fe, Co, Ni, Cu) exhibit stronger electronic synergy due to their multi-directional d-orbital electron distributions. The team proposed electrostatic potential (ESP) as a key descriptor to quantitatively evaluate this synergistic effect, providing a theoretical framework for regulating intermediate adsorption in ORR. The related studies, titled “Dual-Channel σ-Donation/π-Backdonation-Driven p–p Coupling in Calcium Dual-Atom Catalysts Enables Accelerated ORR” and “Electrostatic Potential-Guided Synergy in Atomic Pairs Enables Highly Efficient Oxygen Bifunctional Catalysis,” were published in Advanced Energy Materials and Advanced Functional Materials, respectively. The first authors are Kaiyue Chen and Canhui Zhang, both Ph.D. candidates (Class of 2023), with OUC as the primary corresponding institution.
In parallel, addressing the unclear evolution of active sites and pathway selection in OER, the team constructed a self-adaptive Ni–Co-based MOF-on-MOF catalyst, which undergoes in situ reconstruction into a NiCoOOH active phase with high-density, short-range Ni–Co dual sites during OER. This transformation induces a shift in the OER pathway from the conventional adsorbate evolution mechanism to a lattice oxygen mechanism (Fig. 2). The catalyst requires an overpotential of only 301 mV at 1 A cm⁻² and operates stably for over 1,000 hours. When assembled into an anion exchange membrane alkaline electrolyzer, it achieves stable operation at 1.77 V and 1 A cm⁻² for more than 240 hours.
To further enhance OER activity and interfacial stability under industrial current densities, the team developed a non-precious metal catalyst system based on sulfur–oxygen coordination engineering (SOₓδ⁻–Ni₃S₄@NiFe-MOF). By introducing sulfur–oxygen anions at the interface, the local coordination environment and electronic structure are synergistically optimized, enabling excellent OER activity (1.73 V at 1 A cm⁻²) and long-term stability (over 600 hours) under harsh conditions. The related studies, titled “Dual-Pathway Chloride Suppression Enables Stable Industrial-Scale Anion Exchange Membrane Seawater Electrolysis” and “Unlocking Pathway-Selective OER in a Self-Adaptive MOF-on-MOF Heterostructure toward Industrial-Level Alkaline Water Electrolysis,” were published in Advanced Energy Materials and ACS Catalysis, respectively. The first authors are Xianbiao Hou (Ph.D. candidate, Class of 2021) and Tengjia Ni (Ph.D. candidate, Class of 2024), with OUC as the primary corresponding institution.
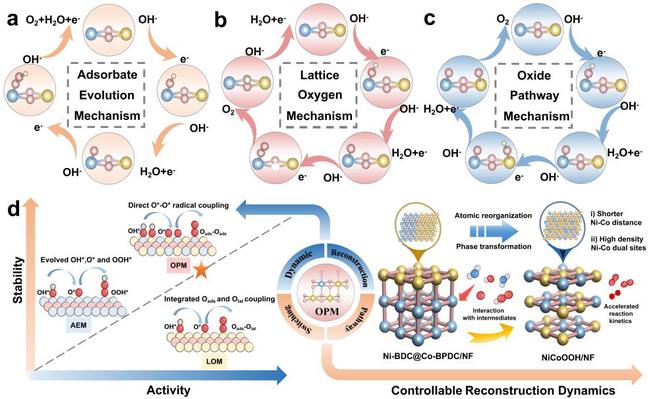
Fig. 2. Relationship between OER dynamic reconstruction and reaction path
These findings advance the understanding of structure–performance relationships and synergistic catalytic mechanisms in non-precious metal oxygen electrocatalysts, providing important theoretical guidance and material support for the development of high-performance energy conversion systems.
Text/Images: Tengjia Ni